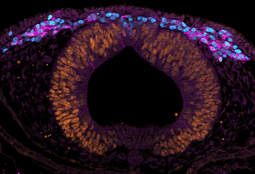
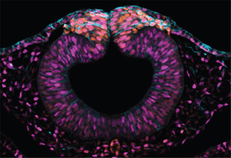
Selected publications
|
Pluripotency of a founding field: rebranding developmental biology. Am Development. Crystal D. Rogers et al., January 2024. https://doi.org/10.1242/dev.202342
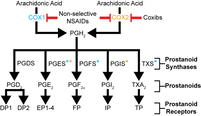
Nonsteroidal anti-inflammatory drugs and implications for the cyclooxygenase pathway in embryonic development. Am J Physiol Cell Physiol. Tess A. Leathers and Crystal D. Rogers. January 2023. https://doi.org/10.1152/ajpcell.00430.2022
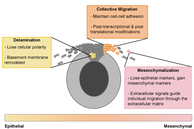
Time to go: neural crest cell epithelial-to-mesenchymal transition. Development. Tess A. Leathers and Crystal D. Rogers. July 2022. https://doi.org/10.1242/dev.200712
The amazing and anomalous axolotls as scientific models. Developmental Dynamics. Carly J. Adamson, Nikolas Morrison-Welch, and Crystal D. Rogers. March 2022. http://anatomypubs.onlinelibrary.wiley.com/doi/10.1002/dvdy.470
|
| |||||||||||||||||||||||||
|
Expression Atlas of Avian Neural Crest Proteins: Neurulation to Migration. Developmental Biology. Brigette Y. Monroy, Carly J. Adamson, Alexis Camacho-Avila, Christian N. Guerzon, Camilo V. Echeverria Jr., Crystal D. Rogers. Accepted, Dec. 2021, Vol. 483, March 2022. https://doi.org/10.1016/j.ydbio.2021.12.018
|
| |||||||
|
Cadherin-11 is required for neural crest specification and survival. Frontiers in Physiology. S. Manohar, A. Camacho-Magallanes, C. Echeverria, Jr., C.D. Rogers. Oct, 2020. https://doi.org/10.3389/fphys.2020.563372
|
| |||||||
|
Early expression of Tubulin Beta-III in avian cranial neural crest cells. Gene Exp. Patterns. J. Chacon, C.D. Rogers. Aug, 2019. doi: 10.1016/j.gep.2019.119067
|
| |||||||
Data on the effects of N-cadherin perturbation on the expression of type II cadherin proteins and major signaling pathways. Data in Brief. C.D. Rogers. Aug, 2018. doi: 10.1016/j.dib.2018.08.029
| Rogers, Aug, DIB. 2018. | |
| File Size: | 1416 kb |
| File Type: | |
A catenin-dependent balance between N-cadherin and E-cadherin controls neuroectodermal cell fate choices. Mechanisms of Development. C.D. Rogers, L.K. Sorrells, M.E. Bronner. July, 2018. doi: 10.1016/j.mod.2018.07.003
| Rogers, Sorrells, and Bronner, 2018 | |
| File Size: | 7782 kb |
| File Type: | |
Specifying neural crest cells: From chromatin to morphogens and factors in between. WIREs Developmental Biology. C.D. Rogers and Nie, S. May 3, 2018. doi: 10.1002/wdev.322
| Rogers and Nie, WIREs Dev Biol, 2018 | |
| File Size: | 3011 kb |
| File Type: | |
Sip1 mediates an E-cadherin-to-N-cadherin switch during cranial neural crest EMT. JCB. C.D. Rogers, A. Saxena, M.E. Bronner. December 2, 2013. doi: 10.1083/jcb.201305050
| Rogers et al., JCB, 2013 | |
| File Size: | 3606 kb |
| File Type: | |
Elk3 is essential for the progression from progenitor to definitive neural crest cell. Dev Biol. C.D. Rogers, J.L. Phillips, M.E. Bronner. February 15, 2013. doi: 10.1016/j.ydbio.2012.12.009.
| Rogers et al., Dev Biol, 2012 | |
| File Size: | 2190 kb |
| File Type: | |
The response of early neural genes to FGF signaling or inhibition of BMP indicate the absence of a conserved neural induction module. BMC Dev Biol. C.D. Rogers, G.S. Ferzli, E.S. Casey. December 15, 2011. doi:10.1186/1471-213X-11-74.
| Rogers et al., BMC Dev Biol, 2011 | |
| File Size: | 619 kb |
| File Type: | |
Neural crest specification: tissues, signals, and transcription factors. WIREs Dev Biol. C.D. Rogers, C.S. Jayasena, S. Nie, M.E. Bronner. November 17, 2011. DOI: 10.1002/wdev.8.
Neural induction and factors that stabilize a neural fate. Birth Defects Res C Embryo Today. C.D. Rogers, S.A. Moody, E.S. Casey. September 2009. DOI: 10.1002/bdrc.20157.
Xenopus Sox3 activates sox2 and geminin and indirectly represses Xvent2 expression to induce neural progenitor formation at the expense of non-neural ectodermal derivatives. Mech of Dev. C.D. Rogers*, N. Harafuji*, T.C. Archer, D.D. Cunningham, E.S. Casey. January-February 2009.
Sox3 expression is maintained by FGF signaling and restricted to the neural plate by Vent proteins in the Xenopus embryo. Dev Biol. C.D. Rogers*, T.C. Archer*, D.D. Cunningham, T.C. Grammer, E.M. Silva Casey. January 2007.